
Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 22.06.2019 06:30
The best solution for preventing harm to people and pets from severe hurricanes involves determining and warning residents about what
Answers: 1

Chemistry, 22.06.2019 15:10
The ozone molecule o3 has a permanent dipole moment of 1.8×10−30 cm. although the molecule is very slightly bent-which is why it has a dipole moment-it can be modeled as a uniform rod of length 2.5×10−10 m with the dipole moment perpendicular to the axis of the rod. suppose an ozone molecule is in a 8000 n/c uniform electric field. in equilibrium, the dipole moment is aligned with the electric field. but if the molecule is rotated by a small angle and released, it will oscillate back and forth in simple harmonic motion.what is the frequency f of oscillation?
Answers: 2
You know the right answer?
How many oxide ions are in 0.55 grams of titanium oxide?...
Questions

Arts, 26.09.2019 10:30





Mathematics, 26.09.2019 10:30

Spanish, 26.09.2019 10:30

Mathematics, 26.09.2019 10:30



Mathematics, 26.09.2019 10:30

Mathematics, 26.09.2019 10:30

Mathematics, 26.09.2019 10:30

Mathematics, 26.09.2019 10:30


English, 26.09.2019 10:30

History, 26.09.2019 10:30


Mathematics, 26.09.2019 10:30

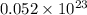 oxide ions
oxide ions 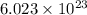 of particles.
of particles.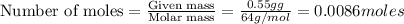
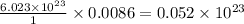 oxide ions
oxide ions 

