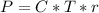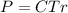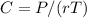Chemistry, 11.06.2020 16:02 bethdove9466
The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute temperature and the gas constant . Suppose the osmotic pressure of a certain solution is measured to be at an absolute temperature o of 312. K. Write an equation that will let you calculate the molarity c of this solution.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1

Chemistry, 23.06.2019 04:00
The movement of tectonic plates and in two locations is described below: location a: tectonic played push together location b: tectonic plates push apart
Answers: 1
You know the right answer?
The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute tempe...
Questions





Social Studies, 23.06.2020 10:57

Mathematics, 23.06.2020 10:57

History, 23.06.2020 10:57



Computers and Technology, 23.06.2020 10:57





Chemistry, 23.06.2020 10:57

Mathematics, 23.06.2020 10:57

Mathematics, 23.06.2020 10:57

Mathematics, 23.06.2020 10:57

Health, 23.06.2020 10:57