CAN SOMEONE HELP ME!!
Solutions of each of the hypothetical acids in the following table are prepared with an initial concentration of 0.100 M. Which of the four solutions will have the lowest pH and be most acidic? Explain please.
Acid pKa
HA 4.00
HB 7.00
HC 10.00
HD 11.00
a. HA
b. HB
c. HC
d. HD
e. All will have the same pH because the concentrations are the same.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1
You know the right answer?
CAN SOMEONE HELP ME!!
Solutions of each of the hypothetical acids in the following table are prepar...
Questions


World Languages, 15.10.2020 19:01


Mathematics, 15.10.2020 19:01

Chemistry, 15.10.2020 19:01

Mathematics, 15.10.2020 19:01



Biology, 15.10.2020 19:01

Mathematics, 15.10.2020 19:01


English, 15.10.2020 19:01


Social Studies, 15.10.2020 19:01


Computers and Technology, 15.10.2020 19:01

Social Studies, 15.10.2020 19:01



Mathematics, 15.10.2020 19:01

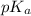 and
and 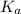 is as follows.
is as follows.