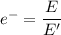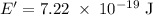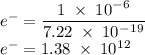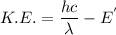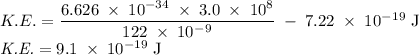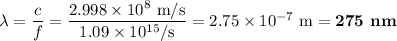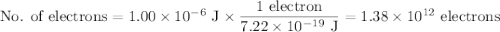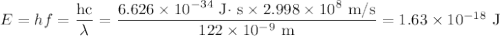Chemistry, 12.06.2020 05:57 coryowens44
Molybdenum metal requires a photon with a minimum frequency of 1.09x1015s-1before it can emit an electron via the photoelectric effect.
a) What is the minimum energy needed to eject an electron?
b)What wavelength of radiation (in nm) will provide a photon of this energy?
c)How many electrons can be freed by a burst of radiation whose total energy is 1.00 μJ, assuming one photon causes one electron to be freed? (μ= micro = 10-6)
d) If molybdenum is irradiated with light of 122nm, what is the maximum kinetic energy of the emitted electrons?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Urea, co(nh2)2, is manufactured on a large scale for use in producing urea-formaldehyde plastics and as a fertilizer. what is the maximum mass of urea that can be manufactured from the co2 produced by combustion of 1.00 x 104 grams of co2?
Answers: 1

Chemistry, 21.06.2019 22:30
Imagine that you’re getting ready to move to a new city. when people move, they are influenced by push factors and pull factors, and you have many reasons for your move. which of the following factors is an example of a pull factor? a. wanting to move because you’ve found a great new school somewhere new b. needing to move because there are not enough resources in your old hometown c. being forced to move because your old home is gone d. having to move because there are no jobs in your current hometown
Answers: 1

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
Molybdenum metal requires a photon with a minimum frequency of 1.09x1015s-1before it can emit an ele...
Questions

Mathematics, 18.10.2019 21:30

Mathematics, 18.10.2019 21:30

English, 18.10.2019 21:30



Mathematics, 18.10.2019 21:30






Mathematics, 18.10.2019 21:30

Mathematics, 18.10.2019 21:30

Mathematics, 18.10.2019 21:30

English, 18.10.2019 21:30

History, 18.10.2019 21:30


Mathematics, 18.10.2019 21:30

History, 18.10.2019 21:30

Chemistry, 18.10.2019 21:30

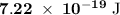 .
.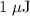 energy has been
energy has been 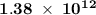 .
.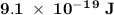 .
.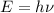
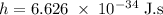
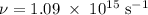
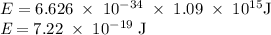
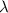 ) of the radiation has been given by:
) of the radiation has been given by: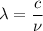
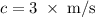
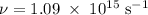 .
.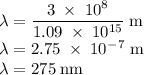
 ) burst out can be given as:
) burst out can be given as: