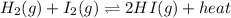Chemistry, 12.06.2020 19:57 UnusualKc5117
Given the following chemical equation at equilibrium: H2(g) + I2(g) ↔2HI(g) + heat. If the temperature is raised, in what direction will the equilibrium shift?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:10
When will le chatelier's principle come into effect? at the beginning of a reaction, when there are only reactants when a reaction has reached chemical equilibrium when a catalyst is added to a reaction mixture when a reaction is occurring but not yet at equilibrium
Answers: 3

Chemistry, 22.06.2019 10:10
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 22.06.2019 22:30
Consider a culture medium on which only gram-positive organisms such as staphylococcus aureus colonies can grow due to an elevated nacl level. a yellow halo surrounds the growth, indicating the bacterium fermented a sugar in the medium, decreasing the ph as a result and changing the color of a ph indicator chemical. this type of medium would be referred to as a differential and enrichment culture.
Answers: 2
You know the right answer?
Given the following chemical equation at equilibrium: H2(g) + I2(g) ↔2HI(g) + heat. If the temperatu...
Questions


History, 30.08.2019 12:30

Social Studies, 30.08.2019 12:30


Geography, 30.08.2019 12:30




Advanced Placement (AP), 30.08.2019 12:30


History, 30.08.2019 12:30


Geography, 30.08.2019 12:30

English, 30.08.2019 12:30

History, 30.08.2019 12:30

Biology, 30.08.2019 12:30

English, 30.08.2019 12:30