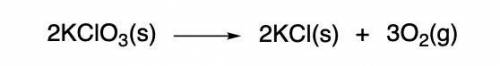
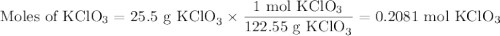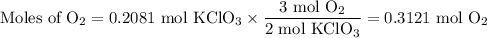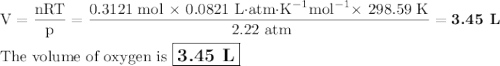I NEED HELP, THANKS! Using the Ideal Gas Law, PV = nRT, where R = 0.0821 L atm/mol K, calculate the volume in liters of oxygen produced by the catalytic decomposition of 25.5 g potassium chlorate according to the following reaction. The oxygen is collected at 2.22 atm and 25.44°C. Express your answer to the correct number of significant figures.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1
You know the right answer?
I NEED HELP, THANKS!
Using the Ideal Gas Law, PV = nRT, where R = 0.0821 L atm/mol K, calculate the...
Questions







Chemistry, 23.06.2019 10:30

Business, 23.06.2019 10:30

Spanish, 23.06.2019 10:30






Mathematics, 23.06.2019 10:30


Advanced Placement (AP), 23.06.2019 10:30

Mathematics, 23.06.2019 10:30

Advanced Placement (AP), 23.06.2019 10:30

History, 23.06.2019 10:30