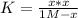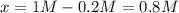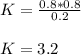Chemistry, 18.06.2020 05:57 andregijoe41
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed in one liter of solution. When equilibrium was established, 0.2 mole of PCl5 remained in the mixture. What is the equilibrium constant for this reaction? (Hint: remember the ICE procedure? initial, change, and equilibrium)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:10
Amonoprotic acid is an acid that donates a single proton to the solution. suppose you have 0.140 g of a monoprotic acid dissolved in 35.0 ml of water. this solution is then neutralized with 14.5 ml of 0.110 m naoh. what is the molar mass of the acid?
Answers: 1

Chemistry, 22.06.2019 04:30
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3
You know the right answer?
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed i...
Questions


English, 01.03.2021 18:50

French, 01.03.2021 18:50






Mathematics, 01.03.2021 18:50


Chemistry, 01.03.2021 18:50



Mathematics, 01.03.2021 18:50

History, 01.03.2021 18:50



English, 01.03.2021 18:50

Mathematics, 01.03.2021 18:50

Mathematics, 01.03.2021 18:50

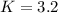
![K=\frac{[PCl_3][Cl_2]}{[PCl_5]}](/tpl/images/0688/6864/bc353.png)
 due to the reaction extent (ICE procedure):
due to the reaction extent (ICE procedure):