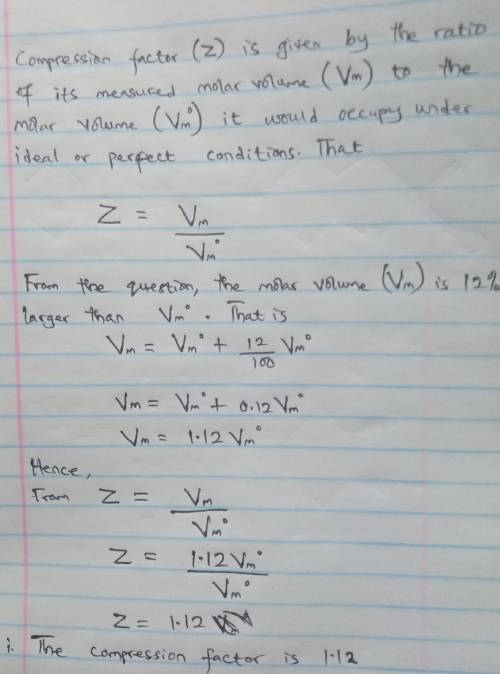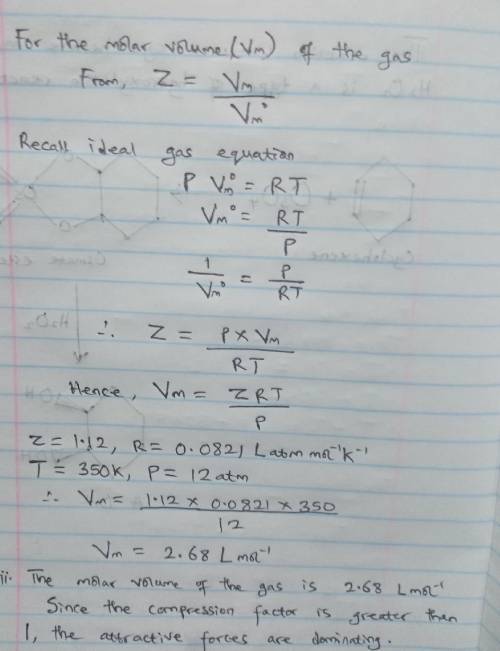A gas at 350 K and 12 atm has a molar volume 12 per cent larger than that calculated from the perfect gas law. Calculate (i) the compression factor under these conditions and (ii) the molar volume of gas. Which are dominating in the sample, the attractive or repulsive forces

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2

Chemistry, 23.06.2019 09:00
Describe the process that was used in this lab to create magnesium oxide, specifically identifying the type of chemical reaction. explain why the product had a higher mass than the reactant, and how this relates to conservation of matter.
Answers: 2
You know the right answer?
A gas at 350 K and 12 atm has a molar volume 12 per cent larger than that calculated from the perfec...
Questions



Biology, 26.02.2021 03:20


Social Studies, 26.02.2021 03:20

Mathematics, 26.02.2021 03:20

English, 26.02.2021 03:20

SAT, 26.02.2021 03:20


Mathematics, 26.02.2021 03:20


Mathematics, 26.02.2021 03:20

Chemistry, 26.02.2021 03:20

Mathematics, 26.02.2021 03:20


English, 26.02.2021 03:20

Mathematics, 26.02.2021 03:20

Mathematics, 26.02.2021 03:20