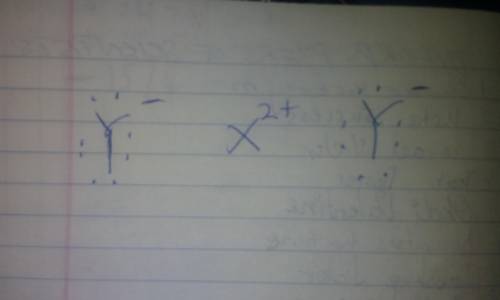Chemistry, 19.06.2020 03:57 rosebelll4555
A metal X loses 2 electrons and a non-metal Y gains 1 electron. Show the electron dot structure of compound formed between them. Is it ionic or covalent? Does it have high or low melting point? Will it conduct electricity in solid state or in aqueous solution and why? Will it be soluble in water?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2

Chemistry, 23.06.2019 00:00
How many atoms or molecules are there in a mole of a substance?
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
A metal X loses 2 electrons and a non-metal Y gains 1 electron. Show the electron dot structure of c...
Questions


History, 19.02.2021 09:30


Biology, 19.02.2021 09:40

English, 19.02.2021 09:40




Mathematics, 19.02.2021 09:40




Physics, 19.02.2021 09:40




English, 19.02.2021 09:40

Mathematics, 19.02.2021 09:40

Mathematics, 19.02.2021 09:40