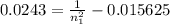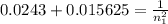Chemistry, 20.06.2020 18:57 tatianaware1617
The electron in a hydrogen atom, originally in level n = 8, undergoes a transition to a lower level by emitting a photon of wavelength 3745 nm. What is the final level of the electron? (c = 3.00 x 108m/s, h = 6.63 x 10−34 J • s, RH = 2.179 x 10−18 J)
a. 5
b. 6
c. 8
d. 9
e. 1

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:30
What must happen before a body cell can begin mitotic cell division
Answers: 1

Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3


Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
The electron in a hydrogen atom, originally in level n = 8, undergoes a transition to a lower level...
Questions


History, 12.02.2021 06:50

Mathematics, 12.02.2021 06:50


English, 12.02.2021 06:50




Mathematics, 12.02.2021 06:50

Mathematics, 12.02.2021 06:50

History, 12.02.2021 06:50


Mathematics, 12.02.2021 06:50

English, 12.02.2021 06:50






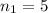
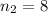
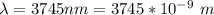
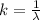
![k = R_{\infty} [\frac{1}{n_1^2} + \frac{1}{n_2^2} ]](/tpl/images/0690/9055/ac88e.png)
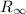 is the Rydberg constant, with a value
is the Rydberg constant, with a value 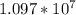
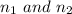 are the principal quantum levels
are the principal quantum levels ![0.0243= [\frac{1}{n_1^2} - \frac{1}{8^2} ]](/tpl/images/0690/9055/cbffd.png)