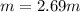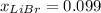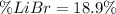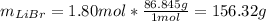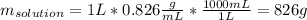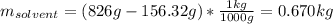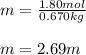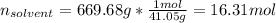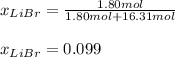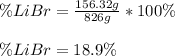Chemistry, 20.06.2020 22:57 shaylawaldo11
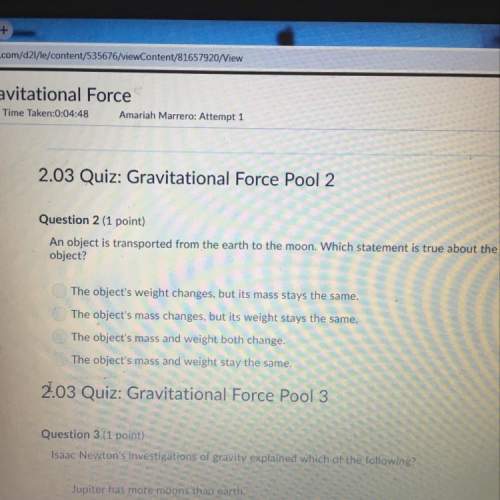
Acetonitrile, CH3CN, is a polar organic solvent that dissolves many solutes, including many salts. The density of a 1.80 M acetonitrile solution of LiBr is 0.826 g/mL. Calculate the concentration of the solution in units of (a) molality; (b) mole fraction of LiBr; (c) mass percentage of CH3CN.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1

Chemistry, 22.06.2019 22:00
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1
You know the right answer?
Acetonitrile, CH3CN, is a polar organic solvent that dissolves many solutes, including many salts. T...
Questions

History, 18.12.2019 06:31


History, 18.12.2019 06:31

Mathematics, 18.12.2019 06:31



Mathematics, 18.12.2019 06:31

Mathematics, 18.12.2019 06:31


History, 18.12.2019 06:31



History, 18.12.2019 06:31

Mathematics, 18.12.2019 06:31



Spanish, 18.12.2019 06:31

Mathematics, 18.12.2019 06:31


Advanced Placement (AP), 18.12.2019 06:31