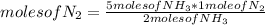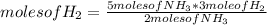N2(g) + 3H2(g) →2NH3(g)
At the end of the chemical reaction, 5 moles of NH3 are produced.
How...

Chemistry, 21.06.2020 22:57 Goldenstate32
N2(g) + 3H2(g) →2NH3(g)
At the end of the chemical reaction, 5 moles of NH3 are produced.
How many moles of N2 and H2 entered the reaction?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
You are to give ampicillin with a recommended dose of 25mg/kg to a child with a mass of 29kg. if stock on hand is 250mg/capsule how many capsules should be given?
Answers: 1

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1

Chemistry, 22.06.2019 19:30
What is the area in square meters of 448 g ai foil that has a thickness of 23921 nm? the density is 2.70 g/cm
Answers: 3
You know the right answer?
Questions


Biology, 20.08.2019 19:30

History, 20.08.2019 19:30

Mathematics, 20.08.2019 19:30




History, 20.08.2019 19:30



Health, 20.08.2019 19:30

Mathematics, 20.08.2019 19:30

History, 20.08.2019 19:30


History, 20.08.2019 19:30

Social Studies, 20.08.2019 19:30

Mathematics, 20.08.2019 19:30


Mathematics, 20.08.2019 19:30