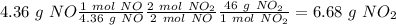Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 4.36 grams of nitrogen monoxide are mixed with excess oxygen gas . The reaction yields 5.46 grams of nitrogen dioxide . nitrogen monoxide ( g ) oxygen ( g ) nitrogen dioxide ( g ) What is the theoretical yield of nitrogen dioxide

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Five students had to answer the question how are elements arranged in a periodic tabledamon: i think the elements are arranged by increasing massflo: i think the elements are arranged according to their properties sienna: i think the elements are arranged by when their discovers kyle: i think the elements are arranged according to how common they areglenda: i don't agree with any of themwho is right
Answers: 1


Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1

You know the right answer?
Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 4.3...
Questions





Biology, 05.07.2019 14:20


Mathematics, 05.07.2019 14:20

Biology, 05.07.2019 14:20

Chemistry, 05.07.2019 14:20

English, 05.07.2019 14:20


History, 05.07.2019 14:20

History, 05.07.2019 14:20

History, 05.07.2019 14:20


World Languages, 05.07.2019 14:20

English, 05.07.2019 14:20


History, 05.07.2019 14:20

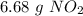
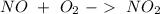
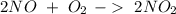
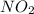 . To do this, we have to first convert the 4.36 g of
. To do this, we have to first convert the 4.36 g of 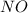 to moles
to moles