
At a certain temperature this reaction follows first-order kinetics with a rate constant of 0.0197s. 2N2O5 (g) right arrow 2N2O4 (g) + O2 (g)Suppose a vessel contains N2O5 at a concentration of 0.280 M. Calculate how long it takes for the concentration of N2Os to decrease by 0.0476 M. You may assume no other reaction is important.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
How many molecules of sucrose c12h22o11 are there in 454 grams of sucrose
Answers: 1

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 10:10
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3

Chemistry, 22.06.2019 18:00
The human activities in two locations are described below: location a: rampant use of plastic containers location b: excessive use of pesticides and fertilizers which statement is most likely true? location a will have poor air quality because plastic is biodegradable. location a will experience water scarcity because plastic absorbs moisture. the population of honeybees will increase in location b because production of crops will increase. the population of fish in location b will decrease because the water is contaminated.
Answers: 1
You know the right answer?
At a certain temperature this reaction follows first-order kinetics with a rate constant of 0.0197s....
Questions


History, 01.12.2020 01:10



Mathematics, 01.12.2020 01:10


Mathematics, 01.12.2020 01:10




Mathematics, 01.12.2020 01:10

English, 01.12.2020 01:10

History, 01.12.2020 01:10

Mathematics, 01.12.2020 01:10




Mathematics, 01.12.2020 01:10


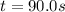
![ln(\frac{[N_2O_5]}{[N_2O_5]_0} )=-kt](/tpl/images/0693/5032/59694.png)
![t=\frac{ln(\frac{[N_2O_5]}{[N_2O_5]_0} )}{-k}=\frac{ln(\frac{0.0476M}{0.280M})}{-0.0197s}\\ \\t=90.0s](/tpl/images/0693/5032/59a2f.png)


