
Chemistry, 26.06.2020 16:01 mathisfun568
4-Methylphenol, CH3C6H4OH (pKa 10.26), is only slightly soluble in water, but its sodium salt, CH3C6H4O-Na , is quite soluble in water. Describe the solubility of 4-methylphenol in solutions of sodium hydroxide, sodium bicarbonate (NaHCO3), and sodium carbonate (Na2CO3). The pKa values for the conjugate acids of sodium hydroxide, sodium bicarbonate (NaHCO3), and sodium carbonate (Na2CO3) are 15.7, 6.36, and 10.33, respectively.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1


Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1
You know the right answer?
4-Methylphenol, CH3C6H4OH (pKa 10.26), is only slightly soluble in water, but its sodium salt, CH3C6...
Questions


Mathematics, 05.11.2019 04:31

Mathematics, 05.11.2019 04:31


History, 05.11.2019 04:31

Biology, 05.11.2019 04:31

English, 05.11.2019 04:31






Physics, 05.11.2019 04:31

Mathematics, 05.11.2019 04:31




Mathematics, 05.11.2019 04:31

Social Studies, 05.11.2019 04:31

Mathematics, 05.11.2019 04:31

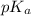 value, weaker will be the acid. Also, an acid completely dissociates into ions in an aqueous base solution when
value, weaker will be the acid. Also, an acid completely dissociates into ions in an aqueous base solution when 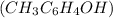 (
(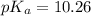 ) is quite soluble in its sodium salt. In NaOH, the dissociation will be as
) is quite soluble in its sodium salt. In NaOH, the dissociation will be as 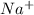 and
and 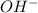 ions as NaOH is a strong base.
ions as NaOH is a strong base.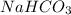 is not a strong base but as 4-Methylphenol forms a sodium salt hence, it will have a low solubility as compared to NaOH.
is not a strong base but as 4-Methylphenol forms a sodium salt hence, it will have a low solubility as compared to NaOH.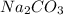 is not a base but when dissolved in water it shows basic character as it produces NaOH (strong base) and
is not a base but when dissolved in water it shows basic character as it produces NaOH (strong base) and 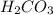 (weak acid). As a result, the solution gets basic. Hence, 4-methylphenol will readily dissolve in
(weak acid). As a result, the solution gets basic. Hence, 4-methylphenol will readily dissolve in 

