Chemistry, 26.06.2020 15:01 ashiteru123
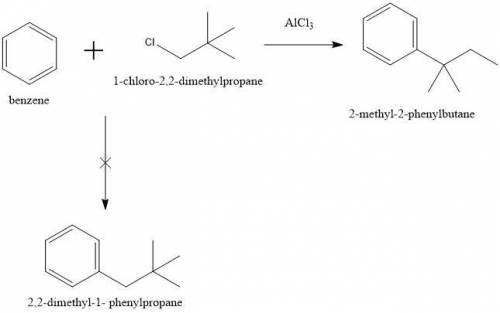
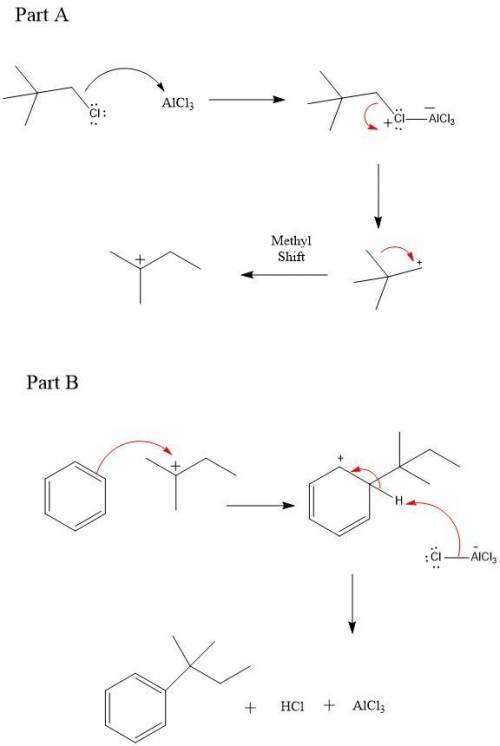
4. When benzene reacts with 1-chloro-2,2-dimethylpropane (neopentyl chloride) in the presence of aluminum chloride, the major product is 2-methyl-2-phenylbutane, not 2,2-dimethyl-1- phenylpropane (neopentylbenzene). Explain this result

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Two nitro no2 groups are chemically bonded to a patch of surface. they can't move to another location on the surface, but they can rotate (see sketch at right). it turns out that the amount of rotational kinetic energy each no2 group can have is required to be a multiple of ε, where =ε×1.010−24 j. in other words, each no2 group could have ε of rotational kinetic energy, or 2ε, or 3ε, and so forth — but it cannot have just any old amount of rotational kinetic energy. suppose the total rotational kinetic energy in this system is initially known to be 32ε. then, some heat is removed from the system, and the total rotational kinetic energy falls to 18ε. calculate the change in entropy. round your answer to 3 significant digits, and be sure it has the correct unit symbol.
Answers: 2

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 10:40
If an area has high air pressure and low humidity, what type of weather will it most likely have? plz !
Answers: 1

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1
You know the right answer?
4. When benzene reacts with 1-chloro-2,2-dimethylpropane (neopentyl chloride) in the presence of alu...
Questions

Mathematics, 18.03.2021 03:20

Chemistry, 18.03.2021 03:20


Mathematics, 18.03.2021 03:20

Mathematics, 18.03.2021 03:20


Mathematics, 18.03.2021 03:20



English, 18.03.2021 03:20



Mathematics, 18.03.2021 03:20



Mathematics, 18.03.2021 03:20




Mathematics, 18.03.2021 03:20

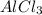 and we form a new bond between the alkyl halide and the
and we form a new bond between the alkyl halide and the