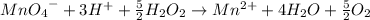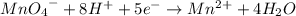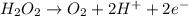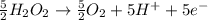Chemistry, 27.06.2020 18:01 nataliemakin7123
The concentration of hydrogen peroxide (H2O2) can be determined by titrating it with an acidified MnO4− solution. The following is an unbalanced equation.
MnO4− (aq) + H2O2 (aq) → O2 (g) + Mn2+ (aq)
(a) Balance the above redox reaction.
(b)Determine the concentration of H2O2 solution in molarity if 10.00 mL of this solution requires 20.00 mL of 1.5 M MnO4− for a complete reaction.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2

Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2

Chemistry, 23.06.2019 03:00
What volume does 1.70 ×10–3 mol of chlorine gas occupy if its temperature is 20.2 °c and its pressure is 795 mm hg?
Answers: 3
You know the right answer?
The concentration of hydrogen peroxide (H2O2) can be determined by titrating it with an acidified Mn...
Questions

Mathematics, 20.02.2021 07:10



Mathematics, 20.02.2021 07:10


Mathematics, 20.02.2021 07:10





Mathematics, 20.02.2021 07:10

Mathematics, 20.02.2021 07:10

Chemistry, 20.02.2021 07:10

Mathematics, 20.02.2021 07:10



Chemistry, 20.02.2021 07:20

Mathematics, 20.02.2021 07:20

Physics, 20.02.2021 07:20