Chemistry, 28.06.2020 02:01 rachiegonzo7
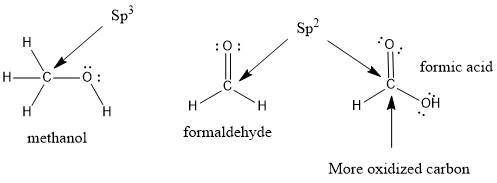
Draw the Lewis structures for CH3OH, CH2O and HCOOH. Indicate the hybrid orbital used in the sigma bonds for each of the carbon atoms. For which of these compounds is the extent of oxidation the greatest? Explain why

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
Draw the Lewis structures for CH3OH, CH2O and HCOOH. Indicate the hybrid orbital used in the sigma b...
Questions


English, 25.10.2019 01:43


Mathematics, 25.10.2019 01:43


Physics, 25.10.2019 01:43


Mathematics, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43








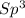 => 4 single bonds
=> 4 single bonds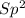 => 1 double bond
=> 1 double bond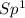 => 1 double bond
=> 1 double bond