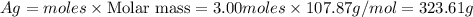Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3
You know the right answer?
What is the mass of silver (107.87 g/mol) produced by the reaction of 3.00 moles of copper with 3.00...
Questions

Social Studies, 31.03.2020 18:10





Chemistry, 31.03.2020 18:11

Physics, 31.03.2020 18:11

English, 31.03.2020 18:11

Computers and Technology, 31.03.2020 18:11

Mathematics, 31.03.2020 18:11

Mathematics, 31.03.2020 18:11

History, 31.03.2020 18:11





Biology, 31.03.2020 18:12


Chemistry, 31.03.2020 18:12

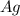 will be produced
will be produced
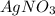 require 1 mole of
require 1 mole of 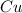
 of
of 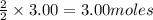 of
of