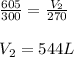Chemistry, 29.06.2020 23:01 kevinleon695
The volume of a gas is 605 liters at 27.0°C. The new temperature is -3.0°C. What is the new volume? (Remember to convert temperatures to the Kelvin scale.)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:00
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 23.06.2019 03:00
In which of the following phases of matter do molecules have the highest amount of energy? a. liquid b. gel c. solid d. gas
Answers: 2

Chemistry, 23.06.2019 13:00
Write the balanced chemical reaction for the formation of fe2(so4)3 from fe2o3 and so3 and determine how many moles of fe2(so4)3 are formed when 12.7 mol of so3 are reacted.
Answers: 1
You know the right answer?
The volume of a gas is 605 liters at 27.0°C. The new temperature is -3.0°C. What is the new volume?...
Questions

Biology, 19.02.2020 16:48




Mathematics, 19.02.2020 16:49



Mathematics, 19.02.2020 16:50



History, 19.02.2020 16:51

Computers and Technology, 19.02.2020 16:51

Mathematics, 19.02.2020 16:51




Mathematics, 19.02.2020 16:52



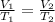
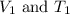 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.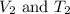 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.