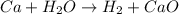Question 4
What is the sum of these two reactions? Write your answer below, but don't worry about formatting with subscripts-Do write chemical formulas with
proper atomic symbols, though (e. g., write CuCl2, not CUCL2).
(1) Ca + 2HCl - CaCl2 + H2
(2) CaCl2+H2O = CaO + 2HCl

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 08:30
7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation. 8. when a 2.5 mol of sugar (c12h22o11) are added to a certain amount of water the boiling point is raised by 1 celsius degree. if 2.5 mol of aluminum nitrate is added to the same amount of water, by how much will the boiling point be changed? show all calculations leading to your answer or use 3 – 4 sentences to explain your answer. 9. if 5.40 kcal of heat is added to 1.00 kg of water at 100⁰c, how much steam at 100⁰c is produced? show all calculations leading to an answer. 10. the freezing of water at 0⁰c can be represented as follows: h2o (l) ↔ h2o(s) the density of liquid water is 1.00 g/cm3. the density of ice is 0.92 g/cm3. in 3 – 4 sentences explain why applying pressure causes ice to melt.
Answers: 1
You know the right answer?
Question 4
What is the sum of these two reactions? Write your answer below, but don't worry about...
Questions

English, 17.12.2020 19:50






History, 17.12.2020 19:50

Mathematics, 17.12.2020 19:50

Mathematics, 17.12.2020 19:50


Mathematics, 17.12.2020 19:50

Health, 17.12.2020 19:50

Mathematics, 17.12.2020 19:50

Computers and Technology, 17.12.2020 19:50

Advanced Placement (AP), 17.12.2020 19:50

Biology, 17.12.2020 19:50

Mathematics, 17.12.2020 19:50


History, 17.12.2020 19:50