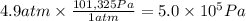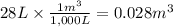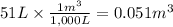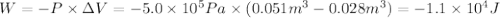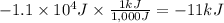Chemistry, 01.07.2020 23:01 thecoolgirl02
Calculate the work (kJ) done during a reaction in which the internal volume expands from 28 L to 51 L against an outside pressure of 4.9 atm. Calculate the work (kJ) done during a reaction in which the internal volume expands from 28 L to 51 L against an outside pressure of 4.9 atm. 11 kJ -11 kJ -39 kJ 39 kJ 0 kJ; No work is done.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Find the mass in grams of hydrogen gas produced when 14.0 moles of hcl is added to an excess amount of magnesium.
Answers: 3

Chemistry, 21.06.2019 16:10
Agas mixture with a total pressure of 745 mmhg contains each of the following gases at the indicated partial pressures: co2, 245 mmhg ; ar, 119 mmhg ; and o2, 163 mmhg . the mixture also contains helium gas. part a what is the partial pressure of the helium gas? phe p h e = nothing mmhg request answer part b what mass of helium gas is present in a 10.2-l sample of this mixture at 283 k ? m m = nothing g request answer
Answers: 1

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

You know the right answer?
Calculate the work (kJ) done during a reaction in which the internal volume expands from 28 L to 51...
Questions



Mathematics, 11.07.2019 03:00


Chemistry, 11.07.2019 03:00

English, 11.07.2019 03:00

Biology, 11.07.2019 03:00



History, 11.07.2019 03:00

English, 11.07.2019 03:00


Social Studies, 11.07.2019 03:00

English, 11.07.2019 03:00

Social Studies, 11.07.2019 03:00





English, 11.07.2019 03:00