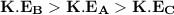Chemistry, 01.07.2020 18:01 twistedhyperboles
Be sure to answer all parts. Three 8−L flasks, fixed with pressure gauges and small valves, each contain 4 g of gas at 276 K. Flask A contains He, flask B contains CH4, and flask C contains H2. Rank the flask contents in terms of:

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
You know the right answer?
Be sure to answer all parts. Three 8−L flasks, fixed with pressure gauges and small valves, each con...
Questions



English, 16.10.2020 14:01

Mathematics, 16.10.2020 14:01

Mathematics, 16.10.2020 14:01

English, 16.10.2020 14:01


Health, 16.10.2020 14:01





History, 16.10.2020 14:01


Mathematics, 16.10.2020 14:01

Health, 16.10.2020 14:01


Mathematics, 16.10.2020 14:01


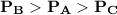
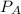 = pressure in the flask A
= pressure in the flask A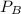 = pressure in the flask B
= pressure in the flask B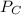 = Pressure in the flask C
= Pressure in the flask C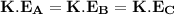
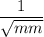
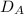 = rate of diffusion in flask A
= rate of diffusion in flask A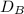 = rate of diffusion in flask B
= rate of diffusion in flask B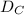 = rate of diffusion in flask C
= rate of diffusion in flask C