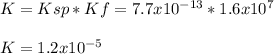Chemistry, 02.07.2020 23:01 graciewilson
Consider the insoluble compound silver bromide , AgBr . The silver ion also forms a complex with ammonia . Write a balanced net ionic equation to show why the solubility of AgBr (s) increases in the presence of ammonia and calculate the equilibrium constant for this reaction. For Ag(NH3)2+ , Kf = 1.6×107 . Use the pull-down boxes to specify states such as (aq) or (s).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1

Chemistry, 22.06.2019 16:00
Which process transfers heat from inside earth to its surface? convection currents in mantle pulling away of tectonic plates drawing in of tectonic plates convection currents in crust
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
Consider the insoluble compound silver bromide , AgBr . The silver ion also forms a complex with amm...
Questions

Mathematics, 28.09.2021 02:10

Physics, 28.09.2021 02:10



Biology, 28.09.2021 02:10



Mathematics, 28.09.2021 02:10


Mathematics, 28.09.2021 02:10

Mathematics, 28.09.2021 02:10


Mathematics, 28.09.2021 02:10







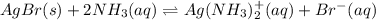
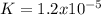
![AgBr(s)\rightleftharpoons Ag^+(aq)+Br^-(aq) \ \ \ Ksp=[Ag^+][Br^-]=7.7x10^{-13}](/tpl/images/0700/1987/811ab.png)
![Ag^+(aq)+2NH_3(aq)\rightleftharpoons Ag(NH_3)_2^+(aq)\ \ \ Kf=\frac{[Ag(NH_3)_2^+]}{[Ag^+][NH_3]^2}=1.6x10^7](/tpl/images/0700/1987/15017.png)
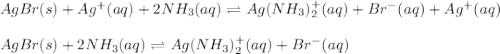
![AgBr(s)+Ag^+(aq)+2NH_3(aq)\rightleftharpoons Ag(NH_3)_2^+(aq)+Br^-+Ag^+\\\\K=[Ag^+][Br^-]*\frac{[Ag(NH_3)_2^+]}{[Ag^+][NH_3]^2}](/tpl/images/0700/1987/20011.png)