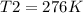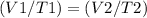Harvey kept a balloon with a volume of 348 milliliters at 25.0˚C inside a freezer for a night. When he took it out, its new volume was 322 milliliters, but its pressure was the same. If the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 23.06.2019 06:50
The student repeated the experiment using a higher concentration of acid. the same volume of acid and the same mass of magnesium ribbon were used. what volume of hydrogen gas would have been produced after 60 seconds?
Answers: 1

Chemistry, 23.06.2019 07:00
In order for a high temperature boiler or steam engine to produce superheated water, or steam: the heat source must be greater than 100°c the water must be permitted to evaporate quickly the system must be sealed and become pressurized above atmospheric pressure the vapor pressure must be kept below 760 mm(hg)
Answers: 1

You know the right answer?
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚C inside a freezer for a night. When...
Questions



English, 11.09.2019 02:30

English, 11.09.2019 02:30

History, 11.09.2019 02:30

Social Studies, 11.09.2019 02:30


Mathematics, 11.09.2019 02:30

English, 11.09.2019 02:30

Mathematics, 11.09.2019 02:30

Mathematics, 11.09.2019 02:30



Social Studies, 11.09.2019 02:30






Mathematics, 11.09.2019 02:30