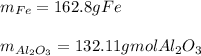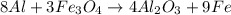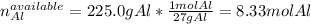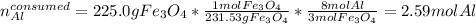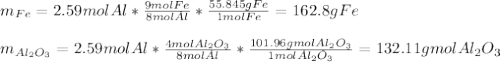Chemistry, 04.07.2020 18:01 DotDotTaterTot
Al + Fe3O4 → Al2O3 + Fe. 13. How many grams of iron are produced by the reaction of 225.0 grams of Al and 225.0 grams of Fe3O4? 14. How many grams of Al2O3 are also produced in the reaction of (23)?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:50
If the mass of the products measured 120g what would the mass of the reactants a. 30g b. 60g c. 120g d. 240g
Answers: 1

Chemistry, 21.06.2019 22:30
Complete the sentence. the lower the hydrogen ion concentration, the the ph. higher lower closer to 7 closer to 0
Answers: 2

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1
You know the right answer?
Al + Fe3O4 → Al2O3 + Fe. 13. How many grams of iron are produced by the reaction of 225.0 grams of A...
Questions





English, 21.07.2019 12:30


Biology, 21.07.2019 12:30

English, 21.07.2019 12:30

Physics, 21.07.2019 12:30





Chemistry, 21.07.2019 12:30





Spanish, 21.07.2019 12:30