
Carbon monoxide (CO) reacts with hydrogen (H2) to form methane (CH4) and water (H20).
CO(g) + 3H2(g) + CH4(g)+H20(9)
The reaction is at equilibrium at 1,000 K. The equilibrium constant of the reaction is 3.90. At equilibrium, the concentrations are as
follows.
[CO] = 0.30 M
[H2] = 0.10 M
[H20] = 0.020 M
What is the equilibrium concentration of CH, expressed in scientific notation?
0.0059
5.9 x 10-2
0.059
5.9 x 102

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2

Chemistry, 23.06.2019 11:20
Match each state of matter with the statement that best describes it.
Answers: 1

Chemistry, 23.06.2019 11:30
A) equal lines b) parallel lines c) perpendicular lines d) none of the above
Answers: 1

Chemistry, 23.06.2019 13:00
Write the balanced chemical reaction for the formation of fe2(so4)3 from fe2o3 and so3 and determine how many moles of fe2(so4)3 are formed when 12.7 mol of so3 are reacted.
Answers: 1
You know the right answer?
Carbon monoxide (CO) reacts with hydrogen (H2) to form methane (CH4) and water (H20).
CO(g) + 3H2(g...
Questions


Mathematics, 25.06.2019 10:00


English, 25.06.2019 10:00

History, 25.06.2019 10:00

Mathematics, 25.06.2019 10:00

English, 25.06.2019 10:00



Mathematics, 25.06.2019 10:00



English, 25.06.2019 10:00


History, 25.06.2019 10:00



Mathematics, 25.06.2019 10:00


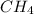 , expressed in scientific notation is
, expressed in scientific notation is 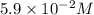
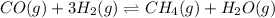
![K_c=\frac{[CH_4]\times [H_2O]}{[CO]\times [H_2]^3}](/tpl/images/0702/2496/1cdb8.png)



