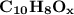Chemistry, 07.07.2020 21:01 guapotaco8102
4.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 128. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured: product carbon dioxide water mass 15.47 g 2.53 g Use this information to find the molecular formula of X.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3


Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1
You know the right answer?
4.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
Questions


Chemistry, 21.04.2021 22:00






Chemistry, 21.04.2021 22:00

Mathematics, 21.04.2021 22:00


Mathematics, 21.04.2021 22:00


World Languages, 21.04.2021 22:00


History, 21.04.2021 22:00



History, 21.04.2021 22:10

Mathematics, 21.04.2021 22:10

History, 21.04.2021 22:10

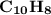 ( Naphthalene )
( Naphthalene )