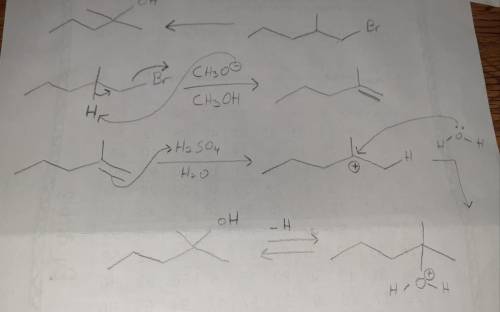
Design a Synthesis of 2-methyl-2-pentanol from 1-bromo-2-methylpentane Part 1: Choose the best option for the immediate precursor to the target molecule. An alkene that has greater substitution on one end of the double bond permits you to control the regiochemistry, placing the OH group selectively on one of the two double bond carbons. This alkene is the best choice, considering the starting material that you have available. Part 2: Choose the best option for the precursor needed to make the alkene. Alkenes can be made in good yield by elimination of HBr from an alkyl halide. Whenever possible, use an alkyl halide that can only make a single alkene by HBr elimination. Part 3: Here is an overview of your retrosynthesis (there is no work, this is just a recap). Part 4 out of 6 Choose the most appropriate reagent(s) for the first step of the synthesis.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 20:30
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1

Chemistry, 23.06.2019 07:00
Introduction of drugs into the gastrointestinal tract is a form of administration. a. enteral b. topical c. parenteral d. inhalation
Answers: 1

Chemistry, 23.06.2019 11:30
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3

Chemistry, 23.06.2019 13:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
Design a Synthesis of 2-methyl-2-pentanol from 1-bromo-2-methylpentane Part 1: Choose the best optio...
Questions



History, 11.02.2020 18:00









Physics, 11.02.2020 18:01




Health, 11.02.2020 18:01



History, 11.02.2020 18:01