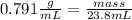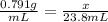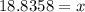Chemistry, 07.07.2020 04:01 thnguyen0720
The density of methanol at 20 degree Celsius is 0.791 g/ml. What is the mass of a 23.8 ml sample of methanol?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1

Chemistry, 23.06.2019 00:30
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1
You know the right answer?
The density of methanol at 20 degree Celsius is 0.791 g/ml. What is the mass of a 23.8 ml sample of...
Questions


Mathematics, 23.01.2021 01:00



History, 23.01.2021 01:00


Mathematics, 23.01.2021 01:00

English, 23.01.2021 01:00

Mathematics, 23.01.2021 01:00



Geography, 23.01.2021 01:00

Mathematics, 23.01.2021 01:00




Mathematics, 23.01.2021 01:00



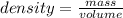
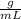 mass= ?volume= 23.8 mL
mass= ?volume= 23.8 mL