Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:10
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 13:00
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

Chemistry, 23.06.2019 00:30
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Answers: 1
You know the right answer?
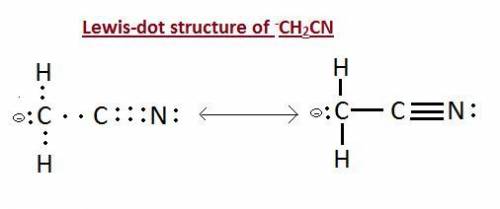
What is the lewis structure for the anion -ch2cn?...
Questions

Mathematics, 18.11.2020 02:20


SAT, 18.11.2020 02:20

Mathematics, 18.11.2020 02:20




English, 18.11.2020 02:20

Mathematics, 18.11.2020 02:20

Mathematics, 18.11.2020 02:20

Mathematics, 18.11.2020 02:20


Mathematics, 18.11.2020 02:20

Mathematics, 18.11.2020 02:20

Mathematics, 18.11.2020 02:20


Mathematics, 18.11.2020 02:20

Chemistry, 18.11.2020 02:20

Chemistry, 18.11.2020 02:20

Mathematics, 18.11.2020 02:20

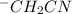 is shown below.
is shown below.