
Chemistry, 08.07.2020 20:01 itsmemichellel
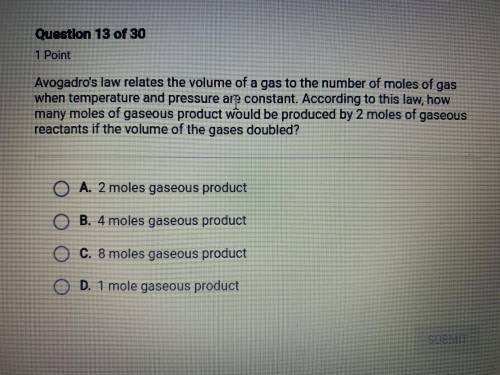
avogadro's law relates the volume of a gas to the number of moles of gas when temperature and pressure are constant. according to this law, how many moles of gaseous product would be produced by 2 moles of gaseous reactants if the volume of the gases doubled?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 21.06.2019 23:00
Which statement describes covalent bases? they have hydroxide ions. they produce hydrogen ions. they are often amines. they are named the same as ionic compounds.
Answers: 3

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2
You know the right answer?
avogadro's law relates the volume of a gas to the number of moles of gas when temperature and pressu...
Questions

Mathematics, 12.05.2021 15:10


Biology, 12.05.2021 15:10


Mathematics, 12.05.2021 15:10





Mathematics, 12.05.2021 15:10


English, 12.05.2021 15:10

Mathematics, 12.05.2021 15:10

Mathematics, 12.05.2021 15:10

Mathematics, 12.05.2021 15:10

Chemistry, 12.05.2021 15:10

English, 12.05.2021 15:10

Mathematics, 12.05.2021 15:10

English, 12.05.2021 15:10




