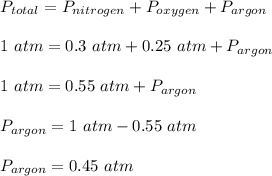Chemistry, 14.07.2020 23:01 smariedegray
The pressure of a 1-L nitrogen gas sample at 25 ⁰C is 0.30 atm. The pressure of a 1-L oxygen gas sample at the same temperature is 0.25 atm. The oxygen gas sample is added to the nitrogen container. Argon gas is added to the mixture until the total pressure of the 1-L container reaches 1.00 atm, and the temperature is adjusted to 25 ⁰C. According to Dalton’s Law of Partial Pressures, the contribution of each gas to the total pressure of the gas mixture is: Nitrogen Oxygen Argon

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Select the correct answer. you have a nightlight plugged into an outlet in the hallway, which uses 3.5 watts when plugged in. if the house circuit provides 120.0 volts, what is the current through this bulb?
Answers: 1

Chemistry, 21.06.2019 16:20
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.
Answers: 2

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1

Chemistry, 22.06.2019 14:00
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2
You know the right answer?
The pressure of a 1-L nitrogen gas sample at 25 ⁰C is 0.30 atm. The pressure of a 1-L oxygen gas sam...
Questions

Mathematics, 01.03.2021 19:30

Social Studies, 01.03.2021 19:30


Mathematics, 01.03.2021 19:30






English, 01.03.2021 19:30


English, 01.03.2021 19:30

Mathematics, 01.03.2021 19:30


Mathematics, 01.03.2021 19:30



History, 01.03.2021 19:30

Physics, 01.03.2021 19:30

Mathematics, 01.03.2021 19:30