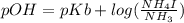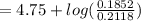Chemistry, 15.07.2020 02:01 kevinvalencia01
An aqueous solution contains 0.397 M ammonia. Calculate the pH of the solution after the addition of 4.63 x 10-2 moles of perchloric acid (HClO4) to 250 mL of this solution. (Assume the volume does not change upon adding perchloric acid). Ka = 5.7 x 10-10, Kb = 1.80 x 10-5

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:40
What is the total reduction potential of a cell in which potassium (k) is reduced and copper (cu) is oxidized? a. 2.59 v b. 3.27 v c. -3.27 v d.-2.59 v
Answers: 1

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2


Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3
You know the right answer?
An aqueous solution contains 0.397 M ammonia. Calculate the pH of the solution after the addition of...
Questions







Mathematics, 19.03.2022 17:00





English, 19.03.2022 17:10


Social Studies, 19.03.2022 17:10

Social Studies, 19.03.2022 17:10

Biology, 19.03.2022 17:20

Mathematics, 19.03.2022 17:20



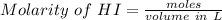
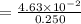
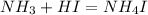
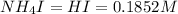
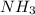 left is
left is