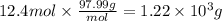Chemistry, 15.07.2020 03:01 anaroles04
If 3.10 moles of P4010 reacted with excess water, how many grams of H3PO4
would be produced?
P4010 +6H20 + 4H3PO4
You Answered
126 g
0 0.007918
Correct Answer
O 1220 g
0.1278
75.98

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Mrs. smith ordered a root beer float (vanilla ice cream + root beer). mrs. smith noticed that the three states of matter (solid, liquid, and gas) all existed simultaneously in her root beer float. a. identify each phase of matter in the root beer float. b. describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? ) c. identify one phase change you would see in a root beer float and described what causes this change.
Answers: 2

Chemistry, 22.06.2019 02:00
For each of the following types of reactions, write a general reaction formula in the symbolic form—for example, a + b → ab. single-displacement double-displacement synthesis decomposition
Answers: 1

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3
You know the right answer?
If 3.10 moles of P4010 reacted with excess water, how many grams of H3PO4
would be produced?
...
...
Questions

Mathematics, 16.12.2020 01:10

English, 16.12.2020 01:10


Mathematics, 16.12.2020 01:10


Mathematics, 16.12.2020 01:10

Mathematics, 16.12.2020 01:10

English, 16.12.2020 01:10


Mathematics, 16.12.2020 01:10


Mathematics, 16.12.2020 01:10

Chemistry, 16.12.2020 01:10

Biology, 16.12.2020 01:10

Mathematics, 16.12.2020 01:10

Mathematics, 16.12.2020 01:10

Mathematics, 16.12.2020 01:10



English, 16.12.2020 01:10