
Chemistry, 15.07.2020 20:01 brapmaster764
an extra strength antacid tablet contains 750 mg of active ingredient, caco3. if it takes 22.25 ml of hcl to neutralize the tablet, how strong is the acid

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1

Chemistry, 23.06.2019 05:20
Explain how global warming could have affected yellowstone frog and salamander habitat's, resulting in changes in the populations of these species
Answers: 2
You know the right answer?
an extra strength antacid tablet contains 750 mg of active ingredient, caco3. if it takes 22.25 ml o...
Questions




History, 13.10.2019 12:10


Health, 13.10.2019 12:10


Computers and Technology, 13.10.2019 12:10


History, 13.10.2019 12:10

Social Studies, 13.10.2019 12:10


Mathematics, 13.10.2019 12:10

Mathematics, 13.10.2019 12:10




Mathematics, 13.10.2019 12:10

History, 13.10.2019 12:10

Chemistry, 13.10.2019 12:10

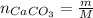
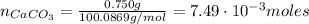
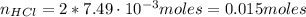
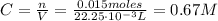
![pH = -log([H^{+}]) = -log(0.67) = 0.17](/tpl/images/0707/3038/32c52.png)


