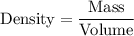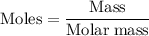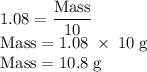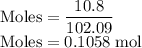Chemistry, 19.07.2020 14:01 robertobi1988
3. The density of acetic anhydride is 1.08 g/mL. How many moles of acetic anhydride are used in this experiment

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

You know the right answer?
3. The density of acetic anhydride is 1.08 g/mL. How many moles of acetic anhydride are used in this...
Questions

Chemistry, 02.05.2021 17:40

Mathematics, 02.05.2021 17:40


Social Studies, 02.05.2021 17:40



Health, 02.05.2021 17:40


Mathematics, 02.05.2021 17:40


Mathematics, 02.05.2021 17:40

Mathematics, 02.05.2021 17:40

Mathematics, 02.05.2021 17:40

Advanced Placement (AP), 02.05.2021 17:40


Physics, 02.05.2021 17:40

Mathematics, 02.05.2021 17:50

Mathematics, 02.05.2021 17:50


Biology, 02.05.2021 17:50