Chemistry, 19.07.2020 17:01 Aurionna101
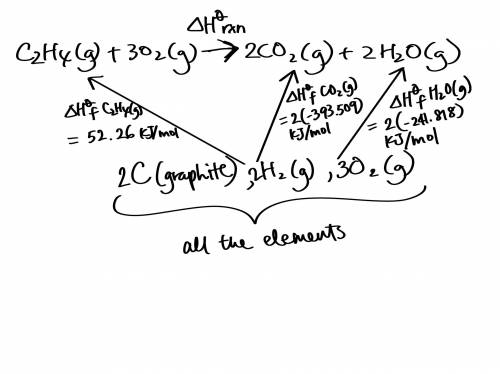
Use the standard enthalpies of formation for the reactants and products to solve for the ΔHrxn for the following reaction. (The ΔHf of C2H4 is 52.26 kJ/mol, CO2 is -393.509 kJ/mol, and H2O is -241.818 kJ.)
C2H4 (g) + 3O2(g) 2CO2 (g) + 2H2O(g)
ΔHrxn = (-345.64 kJ, -583.07 kJ, or -1,322.91 kJ).
The reaction is: (Endothermic or Exothermic).

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 14:30
An object resting on a table weighs 100 n. with what force is the object pushing on the table? with what force is the table pushing on the object? explain how you got your answer.
Answers: 3
You know the right answer?
Use the standard enthalpies of formation for the reactants and products to solve for the ΔHrxn for t...
Questions





History, 18.11.2020 19:00

Biology, 18.11.2020 19:00

Arts, 18.11.2020 19:00





Mathematics, 18.11.2020 19:00



English, 18.11.2020 19:00


Biology, 18.11.2020 19:00


English, 18.11.2020 19:00

Mathematics, 18.11.2020 19:00