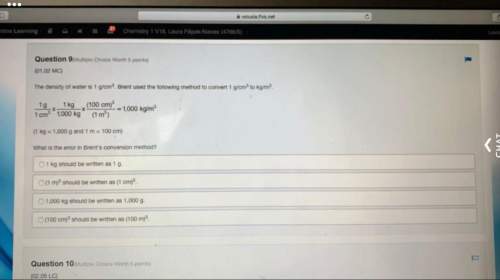
Chemistry, 21.07.2020 14:01 blacksabbath6922
A mixture of reactants and products for the reaction shown below is at
equilibrium in a 5.0 L container. What would most likely happen to the
equilibrium if the volume of the container were reduced to 3.0 L?
N (9) + 3H2(9)
A2NH,
TH (9)
A. The concentrations of N2, H2, and NH3 would decrease until
equilibrium was reached again.
B. More N2, H2, and NH3 would be produced until a new equilibrium
position was reached.
C. More NH3 would be produced until a new equilibrium position was
reached.
D. More Ny and H2 would be produced until a new equilibrium
position was reached.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Motivation cannot be developed with practice; a person either possesses it or they do not.
Answers: 1

Chemistry, 22.06.2019 08:00
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1


Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2
You know the right answer?
A mixture of reactants and products for the reaction shown below is at
equilibrium in a 5.0 L conta...
Questions


Physics, 29.08.2019 18:00




Social Studies, 29.08.2019 18:00

Mathematics, 29.08.2019 18:00



History, 29.08.2019 18:00

Chemistry, 29.08.2019 18:00

Physics, 29.08.2019 18:00



Computers and Technology, 29.08.2019 18:00



History, 29.08.2019 18:00