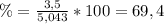1. Unas de las formas de producir nitrógeno gaseoso (N2) es mediante la oxidación de metilamina (CH3NH2), tal como se muestra en la siguiente reacción: CH3NH2 + O2 → CO2 + H2O + N2 Si reaccionan 0,5 mol de metil amina (CH3NH2) con 25,6 g de O2. Determine: a) Balancee la ecuación. (2 ptos) b) ¿Cuántos gramos de nitrógeno (N2) se pueden producir? (4 ptos) c) Si experimentalmente se obtuvieron 3,5 gramos de N2. Determine el porcentaje de rendimiento de la reacción. (4 ptos) Por favor es urgente!!!

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:30
How many joules of heat are absorbed to raise the temperature of 650 grams of water from 5.00c to it's boiling point, 100c
Answers: 1

Chemistry, 21.06.2019 16:00
Endeleev saw trends in the physical and chemical properties of elements when he organized them by
Answers: 2

Chemistry, 21.06.2019 22:00
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1

Chemistry, 22.06.2019 07:20
After watching the video "zinc strip in copper nitrate solution", and reading the instructions, click on the link labeled "start" just below the drawing of the pencil tip. follow the direction to complete the 3x3 grid. answer the below questions for the portion of the activity in which sn(s) is placed in agno3(aq)
Answers: 1
You know the right answer?
1. Unas de las formas de producir nitrógeno gaseoso (N2) es mediante la oxidación de metilamina (CH3...
Questions





Computers and Technology, 21.10.2020 16:01




Social Studies, 21.10.2020 16:01


Computers and Technology, 21.10.2020 16:01









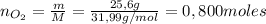
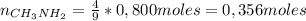
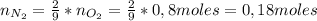
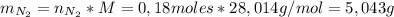
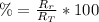
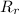 : es el rendimiento real
: es el rendimiento real 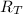 : es el rendimiento teórico
: es el rendimiento teórico