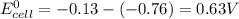Chemistry, 24.07.2020 22:01 bellagracebulle8018
Pb2+(aq) + 2 e- --> Pb(s) Eo = -0.13V Zn2+(aq) + 2 e- --> Zn(s) Eo = -0.76V 9. Given the half-cell potentials above, when the reaction Zn(s) + Pb2+(aq) --> Zn2+(aq) + Pb(s) is made into a voltaic cell, the Ecell is: A. 0.63 V B. -0.63 V C. 0.89 D. 0.89

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which part of a feedback mechanism is able to monitor the conditions outside of cells and usually uses nerve cells to relay this information to an intergrating center
Answers: 2

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2
You know the right answer?
Pb2+(aq) + 2 e- --> Pb(s) Eo = -0.13V Zn2+(aq) + 2 e- --> Zn(s) Eo = -0.76V 9. Given the half-...
Questions

Chemistry, 23.07.2021 14:00





Biology, 23.07.2021 14:00


English, 23.07.2021 14:10

Physics, 23.07.2021 14:10



Physics, 23.07.2021 14:10

World Languages, 23.07.2021 14:10


History, 23.07.2021 14:10

Arts, 23.07.2021 14:10

Mathematics, 23.07.2021 14:10

Mathematics, 23.07.2021 14:10


Physics, 23.07.2021 14:10

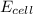 is 0.63 V
is 0.63 V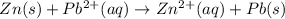
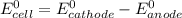
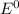 are standard reduction potentials.
are standard reduction potentials.![E^0_{[Pb^{2+}/Pb]}= -0.13V](/tpl/images/0712/6436/bacf1.png)
![E^0_{[Zn^{2+}/Zn]}=-0.76V](/tpl/images/0712/6436/4cd18.png)
![E^0_{cell}=E^0_{[Pb^{2+}/Pb]}- E^0_{[Zn^{2+}/Zn]}](/tpl/images/0712/6436/22253.png)