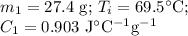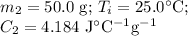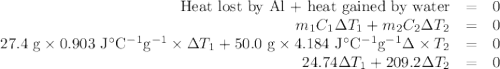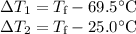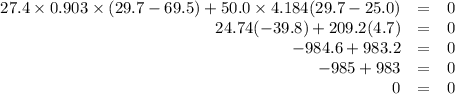Chemistry, 25.07.2020 20:01 kristieroth1
A hot lump of 27.4 g of aluminum at an initial temperature of 69.5 °C is placed in 50.0 mL H2O initially at 25.0 °C and allowed to reach thermal equilibrium. What is the final temperature of the aluminum and water, given that the specific heat of aluminum is 0.903 J/(g·°C)? Assume no heat is lost to surroundings.

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1

Chemistry, 22.06.2019 23:00
What is the measured amount of a product obtained from a chemical reaction?
Answers: 1
You know the right answer?
A hot lump of 27.4 g of aluminum at an initial temperature of 69.5 °C is placed in 50.0 mL H2O initi...
Questions

Physics, 26.03.2021 15:50

Social Studies, 26.03.2021 15:50

Mathematics, 26.03.2021 15:50

Mathematics, 26.03.2021 16:00




Social Studies, 26.03.2021 16:00





Biology, 26.03.2021 16:00

Mathematics, 26.03.2021 16:00




Mathematics, 26.03.2021 16:00