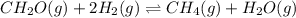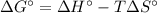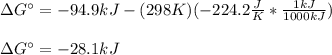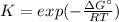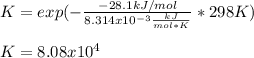Chemistry, 26.07.2020 14:01 dillonmichaeld8069
Calculate the equilibrium constant at 298 K for the reaction of formaldehyde (CH2O) with hydrogen gas using the following information. CH2O(g) + 2H2(g) LaTeX: \longleftrightarrow⟷ CH4(g) + H2O(g) LaTeX: \DeltaΔH°= –94.9 kJ; LaTeX: \DeltaΔS°= –224.2 J/K A. 1.92 B. 9.17 x 10-6 C. 2.07 x 1028 D. 1.10 x 105 E. 8.08 x 104 F. 3.98 x 1011 Group of answer choices

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
An electrons position cannot be known precisely only it's probability of being in a certain location can be known
Answers: 1

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 20:30
Citric acid has a ph between 1 and 3. it is considered to be aa. weak acidb. weak basec. strong based. strong acid
Answers: 2
You know the right answer?
Calculate the equilibrium constant at 298 K for the reaction of formaldehyde (CH2O) with hydrogen ga...
Questions

History, 03.02.2020 01:45

Advanced Placement (AP), 03.02.2020 01:45

Mathematics, 03.02.2020 01:46

English, 03.02.2020 01:46





Business, 03.02.2020 01:46


Mathematics, 03.02.2020 01:46





Mathematics, 03.02.2020 01:46

Mathematics, 03.02.2020 01:46

Social Studies, 03.02.2020 01:46

Mathematics, 03.02.2020 01:46

Biology, 03.02.2020 01:46