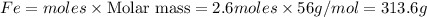Chemistry, 27.07.2020 02:01 stefkellum58
Fe2O3(s) + 3CO(g) ---> 2Fe(l) + 3CO2(g) Steve inserts 450. g of iron(III) oxide and 260. g of carbon monoxide into the blast furnace. After cooling the pure liquid iron, Steve determines that he has produced 288g of iron ingots. Use the theoretical yield of liquid iron and the mass or iron ingots to calculate the percent yield of the reaction.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
The crust of earth may a- continets and ocean floors. b-continents only. c-layers of sedimentary rocks and continents. d-all of the above
Answers: 2

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2

You know the right answer?
Fe2O3(s) + 3CO(g) ---> 2Fe(l) + 3CO2(g) Steve inserts 450. g of iron(III) oxide and 260. g of car...
Questions

World Languages, 08.09.2020 19:01


Physics, 08.09.2020 19:01




English, 08.09.2020 19:01

Arts, 08.09.2020 19:01



Chemistry, 08.09.2020 19:01

Mathematics, 08.09.2020 19:01

Spanish, 08.09.2020 19:01

Biology, 08.09.2020 19:01

English, 08.09.2020 19:01




Geography, 08.09.2020 19:01

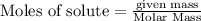
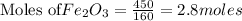
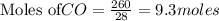
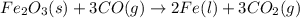
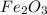 require 3 moles of
require 3 moles of 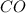
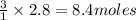 of
of 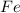
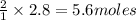 of
of