
Chemistry, 29.07.2020 01:01 drubio102004
A multistep reaction can only occur as fast as its slowest step. Therefore, it is the rate law of the slow step that determines the rate law for the overall reaction. Consider the following multistep reaction:
A+B AB(slow)
A+ABA2B(fast
2A+B A2B(overall)
Based on this mechanism, determine the rate law for the overall reaction.
a) rate = kA2BAB
b) rate = kAB
c) rate = kAAB
d) rate = kA2B

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2
You know the right answer?
A multistep reaction can only occur as fast as its slowest step. Therefore, it is the rate law of th...
Questions

English, 18.01.2020 21:31


History, 18.01.2020 21:31

Chemistry, 18.01.2020 21:31

Biology, 18.01.2020 21:31


Biology, 18.01.2020 21:31


Mathematics, 18.01.2020 21:31


English, 18.01.2020 21:31

Biology, 18.01.2020 21:31



Mathematics, 18.01.2020 21:31

World Languages, 18.01.2020 21:31


English, 18.01.2020 21:31


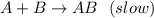
![rate=k[A][B]](/tpl/images/0714/4503/903b2.png)


