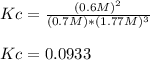Chemistry, 29.07.2020 04:01 sparky1234
The Haber Process is the main industrial procedure to produce ammonia. The reaction combines nitrogen from air with hydrogen mainly from natural gas (methane) and is reversible and exothermic. The enthalpy change for this reaction is - 92 kJ mol-1. In an experiment, 1.5 moles of N2 and 4.0 moles of H2 is mixed in a 1.50 dm3 reaction vessel at 450 °C. After reaching equilibrium, the mixture contained 0.9 mole of NH3.
A) With the above information, write the reaction equilibrium equation in the Haber process. t.
B) Calculate Kc for this reaction.
C) What is the equilibrium yield of ammonia in this reaction?
D) Referring to Le Chatelier's principle and above information, suggest two ways to increase the yield of ammonia in this reaction and explain.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 10:00
Why is the structure of molecule important to its function?
Answers: 1


Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2
You know the right answer?
The Haber Process is the main industrial procedure to produce ammonia. The reaction combines nitroge...
Questions


History, 12.10.2019 00:30

Computers and Technology, 12.10.2019 00:30

Mathematics, 12.10.2019 00:30


Mathematics, 12.10.2019 00:30





Geography, 12.10.2019 00:30

History, 12.10.2019 00:30


Mathematics, 12.10.2019 00:30

Mathematics, 12.10.2019 00:30


Biology, 12.10.2019 00:30



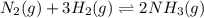 .
.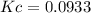 .
.![Kc=\frac{[NH_3]^2}{[N_2][H_2]^3}](/tpl/images/0714/5909/3f3ab.png)
 , we have:
, we have:![[NH_3]=0.6M=2*x](/tpl/images/0714/5909/a9b24.png)
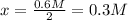
![[N_2]=\frac{1.5mol}{1.5L}-x=1M-0.3M=0.7M](/tpl/images/0714/5909/a34c9.png)
![[H_2]=\frac{4mol}{1.5L}-3*x=2.67M-0.9M=1.77M](/tpl/images/0714/5909/69c53.png)