
Chemistry, 30.07.2020 01:01 pablohc200021
What would be the voltage (Ecell) of a voltaic cell comprised of Cr (s)/Cr3+(aq) and Fe (s)/Fe2+(aq) if the concentrations of the ions in solution were [Cr3+] = 0.75 M and [Fe2+] = 0.25 M at 298K?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 15:00
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
What would be the voltage (Ecell) of a voltaic cell comprised of Cr (s)/Cr3+(aq) and Fe (s)/Fe2+(aq)...
Questions



Mathematics, 16.10.2019 23:00







Business, 16.10.2019 23:00



Computers and Technology, 16.10.2019 23:00


Mathematics, 16.10.2019 23:00




Mathematics, 16.10.2019 23:00

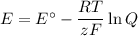
![Q = \dfrac{\text{[Fe}^{2+}]^{3}}{ \text{[Cr}^{3+}]^{2}} = \dfrac{0.25^{3}}{ 0.75^{2}} =\dfrac{0.0156}{0.562} = 0.0278\\\\E = 0.33 - \left (\dfrac{8.314 \times 298}{6 \times 96485}\right ) \ln(0.0278)\\\\=0.33 -0.00428 \times (-3.58) = 0.33 + 0.0153 = \textbf{0.35 V}\\\text{The cell potential is }\large\boxed{\textbf{0.35 V}}](/tpl/images/0715/1287/2ffcc.png)


