Chemistry, 30.07.2020 03:01 christinafish9303
Aspirin (C9H8O4) is produced by the reaction of salicylic acid (C7H6O3, Molar mass = 138.1 g/mol) and acetic anhydride (C4H6O3, Molar mass = 102.1 g/mol) based on the BALANCED equation : C7H6O3(s) + C4H6O3(l ) → C9H8O4(s) + C2H4O2( l) If 63.07 grams of aspirin (Molar mass = 180.2 g/mol) was collected from an experiment when 138.1 grams C7H6O3 reacted with excess C4H6O3, what was the percent yield?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1


Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2
You know the right answer?
Aspirin (C9H8O4) is produced by the reaction of salicylic acid (C7H6O3, Molar mass = 138.1 g/mol) an...
Questions




Mathematics, 18.02.2021 19:00

Chemistry, 18.02.2021 19:00




Mathematics, 18.02.2021 19:00

English, 18.02.2021 19:00


Arts, 18.02.2021 19:00

Computers and Technology, 18.02.2021 19:00

Mathematics, 18.02.2021 19:00

Mathematics, 18.02.2021 19:00


Social Studies, 18.02.2021 19:00


Arts, 18.02.2021 19:00

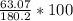 hould be used up in the reaction, yielding 180.2 g of C9H8O4. ⇒ Theoretical yield = 180.2g
hould be used up in the reaction, yielding 180.2 g of C9H8O4. ⇒ Theoretical yield = 180.2g