Chemistry, 30.07.2020 04:01 jadkins842
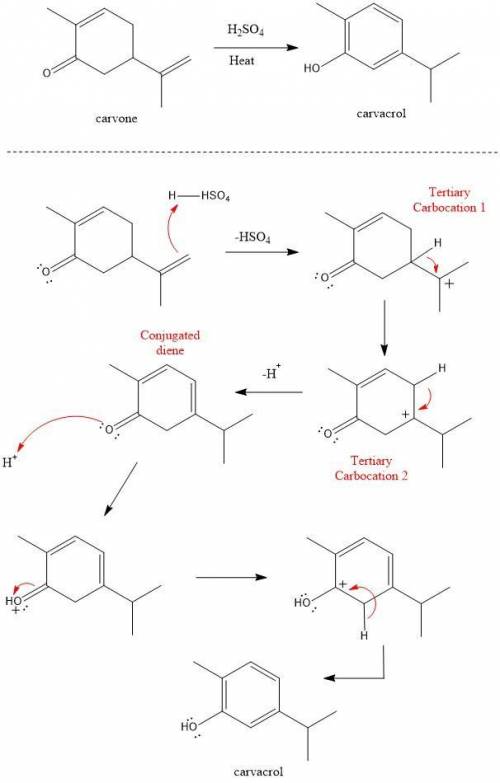
Heating carvone with aqueous sulfuric acid converts it into carvacrol. The mechanism involves the following steps:
1. The terminal alkene of carvone reacts with acid to form tertiary carbocation 1;
2. A hydride shift results in the formation of tertiary carbocation 2;
3. Deprotonation of the ring leads to conjugated diene 3;
4. Deprotonation at the α carbon leads to the product carvacrol.
Required:
Draw the mechanism and then draw the structure of tertiary carbocation 2.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
What approach is required to balance the objectives of sustainable development? balancing the objectives of sustainable development requires a(n) .
Answers: 3

Chemistry, 22.06.2019 04:00
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3

Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
Heating carvone with aqueous sulfuric acid converts it into carvacrol. The mechanism involves the fo...
Questions


Computers and Technology, 12.11.2019 20:31





Social Studies, 12.11.2019 20:31











Biology, 12.11.2019 20:31