Chemistry, 30.07.2020 08:01 putaprincess16
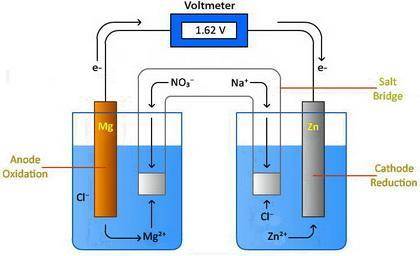
Design a voltaic cell using magnesium as one of the electrodes. Magnesium can be represented as either Metal A or Metal B in the above drawing. Use metal chlorides as the solutions in the two chambers. For example, magnesium chloride, (MgCl2) will be in solution in the chamber with the magnesium electrode. Use NaNO3 in the salt bridge. Select another element for the other electrode. Explain why you selected this element. Include information about the activity of the metal you select and the need for a spontaneous reaction. Metal A: Metal B: In the drawing, 1. Label the oxidation compartment: 2. Label the reduction compartment. 3. Label the direction of the flow of electrons. 4. Label the flow of the magnesium ions. 5. Label the flow of your selected element's ions. 6. What is leaving the salt bridge in the anode compartment? 7. What is leaving the salt bridge in the cathode compartment? 8. Write the oxidation and reduction half-reactions. 9. Calculate the chemical potential of your cell. Show all of your work.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 10:00
Why is the structure of molecule important to its function?
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2
You know the right answer?
Design a voltaic cell using magnesium as one of the electrodes. Magnesium can be represented as eith...
Questions

English, 24.04.2020 22:23

Computers and Technology, 24.04.2020 22:23


Mathematics, 24.04.2020 22:24


English, 24.04.2020 22:24

Physics, 24.04.2020 22:24




Mathematics, 24.04.2020 22:24




History, 24.04.2020 22:24


Mathematics, 24.04.2020 22:24

Mathematics, 24.04.2020 22:24


Mathematics, 24.04.2020 22:24